If you work in nutraceuticals, you’ve probably noticed something:
“Liposomal” has been one of the top keywords over the past couple of years.
But here’s the honest truth — not all liposomal products are built the same. Some are carefully engineered nano-delivery systems. Others are simply phospholipid blends with impressive marketing language.
As the category grows, clarity matters.
Let’s break down what real liposomal technology actually means — and why structure, validation, and engineering matter more than the label itself.
What a True Liposome Actually Is
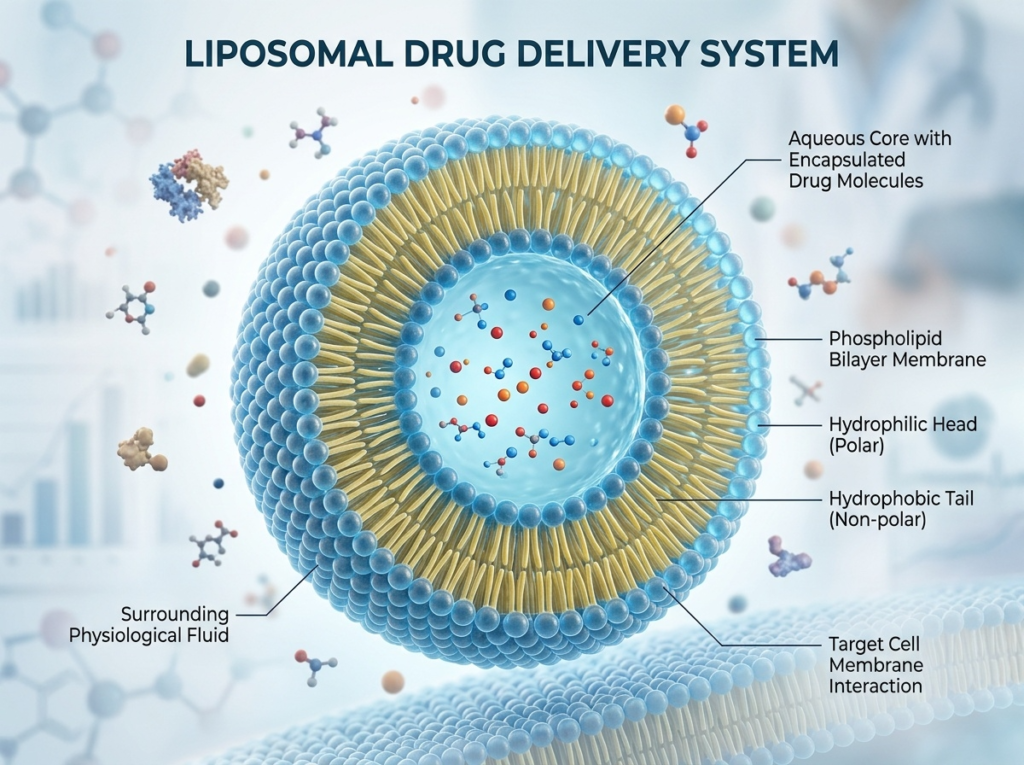
A liposome is a nanoscale vesicle formed from phospholipid bilayers — the same material that makes up our cell membranes.
Each phospholipid has:
- a hydrophilic (water-loving) head
- a hydrophobic (water-repelling) tail
When placed in water, they self-assemble into a bilayer shell, forming a microscopic “bubble” with an aqueous core inside.
This architecture allows:
- Hydrophilic actives (like Vitamin C) to be encapsulated in the core
- Lipophilic compounds to integrate within the lipid membrane
- Protection against oxidation and environmental degradation
- Interaction with biological membranes for potential absorption support
But forming vesicles is only step one. The real challenge is maintaining structural integrity, stability, and reproducibility.
Why the Market Feels Confusing
Today’s “liposomal” products generally fall into three categories:
A) Conventional Liquid Liposomes
Produced through lipid hydration and high-pressure homogenization.
Strengths
- Established technology
- Works well in liquid formats
Limitations
- Aggregation or vesicle fusion over time
- Burst release of hydrophilic actives
- Difficult conversion into stable powder formats
B) Lecithin Blends Marketed as Liposomal
Some formulations simply mix phospholipids with a high percentage of active ingredient.
They may look cloudy in water, but:
- Vesicle formation is often unverified
- DLS particle size data may be unavailable
- Encapsulation efficiency is not demonstrated
- Turbidity ≠ confirmed liposome structure
This is where much of the confusion begins.
C) Engineered Powder Liposomal Systems
The most technically demanding category involves systems designed to:
- Maintain nano-scale structure
- Survive spray drying
- Reconstitute into stable vesicles
- Demonstrate controlled release characteristics
These systems typically target:
- Particle size ~120–200 nm
- Zeta potential beyond ±30 mV
- Verified encapsulation
- Stability under accelerated conditions
Powder liposomes are particularly challenging because drying can cause:
- Vesicle collapse
- Oxidation
- Aggregation upon rehydration
Engineering solutions must address each of these risks.
The 5 Structural Tests That Matter
If someone claims an ingredient is liposomal, here are the questions worth asking:
✔ DLS (Dynamic Light Scattering) Does the particle size fall within a narrow nano range?
✔ Zeta Potential Is the surface charge strong enough to prevent aggregation?
✔ Morphology (TEM/SEM) Are vesicular structures clearly visible?
✔ Encapsulation Efficiency Has free vs. encapsulated active been separated and quantified?
✔ Stability & Release Data Does the structure remain intact under storage and physiological conditions?
Without these, “liposomal” becomes descriptive language — not structural confirmation.
Bioavailability: When Structure Translates to Performance
One of the reasons liposomal delivery attracts so much attention is absorption.
In a controlled human study comparing a structured liposomal Vitamin C system to conventional Vitamin C, results showed:
- 7.62× higher AUC
- 6.29× higher Cmax
- Extended half-life
This demonstrates that properly engineered liposomal systems can produce measurable differences in systemic exposure.
However, it’s important to remember:
Not every product labeled “liposomal” will achieve these outcomes.
Structure drives performance — not terminology.
Why “95% Liposomal Vitamin C” Claims Don’t Make Sense
Occasionally, powders are marketed as “95% liposomal Vitamin C.”
From a material science perspective, this raises questions.
A true liposome requires sufficient phospholipid mass to construct bilayer membranes. If 95% of a powder is active, only 5% remains for:
- Phospholipid structure
- Stabilizers
- Protective matrix components
At that ratio:
- Bilayer formation becomes physically limited
- Encapsulation capacity is constrained
- Powder reconstitution stability is unlikely
- Much of the active would remain unencapsulated
In many such cases, the formulation may be a lecithin blend rather than a verified vesicular system.
True liposomal powders typically require a meaningful proportion of structural material to maintain integrity.
Final Thoughts: Liposomal Is an Engineering Discipline
As the category matures, the differentiator will no longer be the word “liposomal.”
It will be:
- Structural validation
- Stability engineering
- Encapsulation integrity
- Demonstrated performance
Liposomal technology works — but only when structure defines function.
For formulators, brand owners, and sourcing teams, the more relevant question today is not:
“Is it liposomal?”
It is:
“Is it structurally validated, reproducible, and performance-supported?”
Because in advanced delivery systems, science — not labels — is what ultimately earns trust.
#Liposomal #LiposomalTechnology #DrugDelivery #NanoDelivery #Nanotechnology #Encapsulation #Phospholipids #Bioavailability